Ensuring only compliant products get to market. Fast!
Key Takeaways
- Regulatory compliance must be supported earlier in the product lifecycle to ensure products can be developed, produced, and distributed to their target markets faster.
- Compliance, which is often tightly coupled with other regulatory requirements like labeling or permitting, varies by industry, country, and product.
- Best-in-class compliance management solutions leverage commercial databases and support custom rules that validate product information as it is created minimizing waste, streamlining timelines, lowering costs, and improving time-to-market.
- Aras’ digital thread solution enables proactive compliance management for development, automates documentation for compliance audits, and supports advanced capabilities such as integration to an AI-based platform for managing requirements.
Introduction
Getting products to market faster is a common goal in most organizations. The benefits of doing this are well established, but execution is hard. Ensuring that all essential procedures are completed to satisfy product standards can be a meticulous and lengthy process. Unfortunately, all it takes is a single missed compliance requirement and a product can become non-saleable. As an example of the consequences, a beverage company CIMdata worked with told the story of a supplier change that turned finished, ready-to-ship product into scrap. The new supplier did not have a Kosher certification for its ingredient, but the product it went into was certified Kosher. The beverage company realized the problem only after the product was manufactured and ready for shipment. Once the issue was identified, the product could not be sold and had to be scrapped. On the plus side, they avoided a recall, but all the investment such as working capital and production capacity was lost.[1]
The Food and Beverage industry is not the only place with regulations. Some of the regulatory governing bodies in different domains include:
- Industry—US Federal Aviation Administration (FAA), US Food and Drug Administration (FDA), United States Department of Agriculture (USDA), European Union Aviation Safety Agency (EASA), European Medicines Agency (EMA), and European Food Safety Authority (EFSA)
- Safety—US Occupational Safety and Health Administration (OSHA), European Agency for Safety and Health at Work (EU-OSHA), US Centers for Disease Control and Prevention (CDC), European Centre for Disease Prevention and Control (ECDC), and US National Institute for Occupational Safety and health (NIOSH)
- Emissions—US Environmental Protection Agency (EPA), and European Environment Agency (EEA)
- Climate/Green—US Environmental Protection Agency (EPA), US Department of Energy (DOE), US Federal Trade Commission (FTC), and European Union Agency for the Cooperation of Energy Regulators (ACER)
There are many others that impact many industrial sectors. Clearly, regulatory bodies and evolving regulations are everywhere. Most recently, there is an emergence of new compliance requirements coming from Europe that will eventually impact most product organizations.
The European Union is spearheading a transformative initiative, the Digital Product Passport (DPP), which is integral to its Sustainable Product Regulation proposal and the European Green Deal. This innovative data exchange framework is designed to encapsulate information on product sustainability, environmental impact, and recyclability. Its primary goal is to enhance transparency and efficiency throughout supply chains, thereby bolstering product sustainability and championing the principles of a circular economy within the EU. The concept of a circular economy, as articulated by the Ellen MacArthur Foundation, has long been advocated by CIMdata. The DPP's reach extends well beyond European borders, necessitating compliance from suppliers and manufacturers worldwide to meet its stringent requirements if they want to sell products within the EU.
To comply with regulations, companies developing products treat regulations as requirements and incorporate them into specifications that describe the requirements that must be satisfied to deliver the product to customers. Ideally, requirements management solutions are used to perform this task, but it is often done in Microsoft Office documents (Excel, PowerPoint, and Word) or siloed tools. Product planning, regulatory affairs, compliance, and quality teams are siloed in function and the tools they use to manage requirements. The complexity of development tasks such as design, simulation, and testing increase because the requirements are dispersed across various repositories, each with its unique interface. Due to this complexity, verification tends to happen late in the development cycle leading to expensive surprises when requirement misses are identified.
A further complication faced by companies is that regulations are not static. They evolve and can affect existing products. Companies need a robust way to assess the compliance of current products with new regulations.
Compliance Management Best Practices
 As with other aspects of implementing PLM, eliminating silos by connecting data and processes generates value. Compliance can be compared to a dumbbell in terms of its lifecycle workload distribution. Initially, there is a significant effort to pinpoint the necessary compliance standards. Subsequently, towards the end of the lifecycle, extensive verification and validation are undertaken to confirm that the product fulfills these established requirements. In compliance management, it's crucial to advance or “shift left” the verification and validation processes to an earlier stage in the product lifecycle. By doing so, potential issues can be identified and resolved promptly, which helps minimize unexpected costs and delays.
As with other aspects of implementing PLM, eliminating silos by connecting data and processes generates value. Compliance can be compared to a dumbbell in terms of its lifecycle workload distribution. Initially, there is a significant effort to pinpoint the necessary compliance standards. Subsequently, towards the end of the lifecycle, extensive verification and validation are undertaken to confirm that the product fulfills these established requirements. In compliance management, it's crucial to advance or “shift left” the verification and validation processes to an earlier stage in the product lifecycle. By doing so, potential issues can be identified and resolved promptly, which helps minimize unexpected costs and delays.
In addition to shifting left, validation should happen in real-time. As a developer is inputting data into a model whether geometry, material, tolerance, or any other attribute, a tool should be consuming requirements and assessing the data input within the design context to ensure it is compliant. If it is not compliant, the data should be flagged, and recommendations made. Real-time feedback enables the developer to address issues while they are top of mind.
Integrating compliance and risk management with product data and processes forms what's known as the digital thread, powered by a product innovation platform.[2] This concept, extensively covered by CIMdata, hinges on the traceability of information through interconnected data. Such an environment allows for functionalities like change impact analysis, where a modification in a requirement, like an emission value, reveals all related components—ranging from parts to production plans. This traceability not only aids in informed decision-making but also provides a retrospective path to trace a product's history from conception to market, proving invaluable for resolving product challenges.
Compliance audits are an inevitable part of business operations, which can be daunting for companies. When data is connected and navigable, as in Aras Innovator, searches and reports can be easily developed or created on the fly to ensure auditors get the information required. Figure 1 shows a high-level view of what a left-shifted compliance process looks like in a modern manufacturing environment.
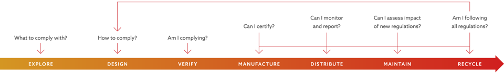
Figure 1 - Holistic View of how Compliance Management integrates with the Product Lifecycle
(Courtesy of Aras)
To make this concept work, regulatory compliance must be integrated with processes across the enterprise, partners, and supply chain, i.e., via a digital thread. To create an effective digital thread a product innovation platform is required to manage data and processes to enable traceability.
Aras’ Approach to Compliance Management
Aras is a leading PLM and digital thread solution provider offering extensive support for various aspects of compliance management. Material data is a core attribute of part items. The data model can be extended to support additional commercial or proprietary attributes. Compliance checks can be configured into workflows to ensure compliance when parts and BOMs reach appropriate lifecycle states. Out-of-the-box, Aras provides support for material-based compliance requirements such as REACH and RoHS. BOM reports can show a color-coded report with compliance status.
As products become more electrified and software-driven, electronics content becomes core. Items within Aras Innovator can represent mechanical, electrical, electronic, software, and process industry items such as substances, ingredients, and recipes. Aras has had an integration into the IHS CAPS Universe database within the Aras Component Engineering solution[3] for many years that contains a wealth of compliance data including REACH, RoHS, Certificates of Conflict Mineral compliance, and other important data needed to properly manage components.
Product and performance requirements are managed as structures of items within the Requirements Management solution and can be related to other structures as show in Figure 2 or in a model-based systems engineering (MBSE) context.
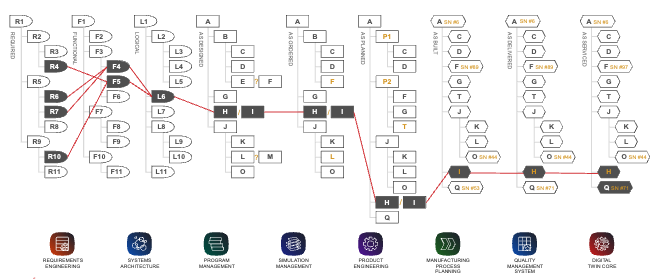
Figure 2 – A Structure-View of the Product Lifecycle
(Courtesy of Aras)
The relationships between items and structures form the digital thread. This linkage along with navigation and reporting capabilities makes understanding compliance status a simple process. Digital thread compliance information is viewed in context of the overall product and development project improving decision making. CIMdata believes that accessing high-quality information fast is critical to enabling enterprise level innovation.
Aras’ collaboration capabilities can be configured to support supplier, development partner, supply chain, and customer access as a holistic, secure ecosystem. Aras Innovator SaaS simplifies access control, and web services including SOAP, REST, and commercial integration technology support more complex connectivity. Aras recently released its Aras Supplier Management Solutions[4] which includes access to the via the internet-facing Aras Portal ensuring that the extended ecosystem only accesses what they are supposed to access. Key capabilities of the solution include:
- Track Supplier Information, Contacts, and Monitoring
- Collaboration with threaded discussions
- Surveys and Performance Cards
- CAD/Part/Document Sharing
- Corrective Action and Negotiation Requests
- Aras Portal with mobile or desktop web interface
All data is connected via Aras’ digital thread and can be configured to meet any special requirements via low code support. Additionally, Aras has long been known for its ability to upgrade any customer’s configuration at no additional cost beyond the base subscription.
Aras, like most other solution providers, has an Artificial intelligence (AI) strategy. Aras’ open data model and ease of integration make it an easy solution for 3rd parties to connect with. Within the compliance space, Fraunhofer an Aras partner is developing a solution that integrates with Certivity’s Regulation Management database. Published regulations and proprietary standards are processed and managed and exposed in a consumable format. Product developers can find appropriate regulations that must be supported and the translated requirements are linked to Aras Innovator via ReqIF. The transformation of regulatory documents into consumable requirements is a huge help to product planning and development processes.
Customers
Compliance support with Aras Innovator is not just theoretical; several customers delivered compliance-related presentations at the recent ACE 2024 event.[5] SICK, a global manufacturer of industrial sensors uses Aras Innovator to ensure compliance with green regulations including CO2 output by encoding regulations in requirements and validating products against those requirements.
Edwards Lifesciences, known for their expertise in cardiac devices, has adopted the Aras platform to streamline global regulatory compliance and distribution. The platform effectively oversees the entire lifecycle of a product, from production to global device registration, while also keeping track of post-market performance. This process ensures that products meet specific market criteria before they are sold. By continuously updating product certifications, Edwards can dynamically adjust their “Allowed to Sell” status, enhancing inventory control and minimizing the likelihood of recalls due to regulatory issues. CIMdata sees this customer-developed solution as a great example of leveraging compliance information using a low code development approach.
Conclusion
Regulatory compliance is a critical process in companies. Poor management of the process can negatively impact quality, cost, and time-to-market. Companies that sell products into global markets must ensure compliance continuously to avoid expensive surprises at launch or even post-launch. Converting regulations into requirements requires specialists in the regulatory domain. Best-in-class PLM solutions provide a platform to enable incorporation of necessary regulatory domains. Aras Innovator is one of those solutions that has a long history of enabling disparate database integration and digital thread enablement. Companies looking to apply the shift left strategy to regulatory compliance processes should evaluate Aras Innovator.




