Key takeaways:
- Complete and accurate design history files (DHF) and device master records (DMR) are critical to successfully launching and supporting medical device products
- Paper or file folder-based processes often used to manage documentation slow time to market, make data hard to manage, and make effective risk management, verification, validation, and full traceability difficult to support
- Minerva’s Medical Device solution, based on Aras Innovator, helps companies develop and support products by structuring data and processes using industry best practices to speed time to market while managing risk
Introduction
The medical device industry consists of products ranging from single components such as a bone screw to complex diagnostic systems that can include mechanical, electronic, software, and formulated components. Quality and compliance processes are critical to ensuring product safety and are monitored closely by regulatory agencies. Time to market is key to business success. Within our consulting practice, the common issues for medical product companies seen by CIMdata include:
- Disconnected data stored in many spreadsheets or standalone repositories and file stores
- Complex and slow approval processes
- Incomplete traceability
Disconnected data impacts both quality and speed. Multiple data repositories must be searched independently and search results interpreted and manually connected. This is error prone and risky because it may not be clear that all the appropriate data has been identified and if it is the latest version of the data. This problem can ultimately lead to poor decisions and errors from using incorrect data.
Complex and slow approvals increases elapsed time. Based on CIMdata’s consulting experience, when there are more than 3 or 4 approvers, the additional approvers do not add much, if any value. The additional approvers tend to approve based on who previously approved, or if a key influencer has approved the item.
The U.S. Food and Drug Administration (FDA) and other medical device regulatory bodies require that the device master record (DMR) contains a complete, accurate documentation set required to produce a device, and that the DHF contains the supporting documentation for product decisions traceable from requirements and claims, through verification and validation.
These issues cause many problems that add risk and slow time to market. By implementing a solution to address these issues a medical device manufacturer can improve margins, time to market, quality, compliance and, perhaps most important, reduce overall business risk.
About Minerva
The Minerva Group, headquartered in Denmark is a global supplier of solutions and support services to manufacturing companies. Founded in 1995, Minerva initially focused on enterprise resource planning (ERP) and expanded to support product lifecycle management (PLM) in 2000. Since 2000 they have helped over 70 companies improve their operations by implementing PLM solutions. Key customers include Airbus, Saab AB, Lockheed, Carestream, and Aeronamic.
The Minerva Group has been a leading solution developer and implementation partner for Aras Corp since 2008. Aras is the developer of the Aras Innovator PLM solution, known for its innovative solution modeling environment and business model. Minerva uses their implementation experience to develop industry-focused solutions that leverage the underlying capabilities and flexibility of the Innovator platform. Specifically, Minerva offers solutions to support high-tech electronics and medical devices.
Minerva’s Medical Device Solution
The Minerva Medical Device PLM solution supports key medical device developer needs including creating and managing the DHF and DMR, and supporting pro-active Risk Management. CIMdata views industry-specific templates and solutions as an effective way to speed up PLM implementations and shorten time to value. Minerva’s templating capabilities, shown in Figure 1, allow both the structure and content of the DHF and DMR to be defined and managed. A deliverable matrix template associates file document templates, including Microsoft Office documents, with the DMR and DHF structure allowing users to traverse a single logical source of truth which makes data a competitive advantage. By leveraging the versioning capabilities of the underlying Aras platform, Minerva combines the structures and documents into a versionable project. Tailored workflows capture approvals for minor and major changes as well as lifecycle state changes. Project plans often change over time as a project progresses and companies need to keep track of what changed in the project plan, and why, to fully address regulatory requirements. CIMdata believes that versionable projects are a straightforward way to capture this key element of traceability.
Mr. David Sherburne, Executive IT Director at Carestream Health, is a long time Aras customer, and has presented at several Aras ACE conferences. Carestream is using the Minerva solution within R&D. He stated that “it works well, captures product documentation holistically, not just to support regulatory needs. The solution has replaced many point solutions and provides really strong historical data for documents and projects.”
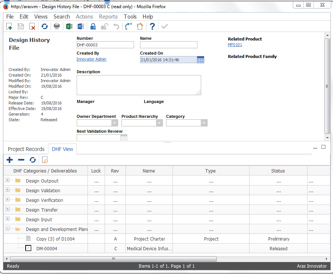
Figure 1—Template for Configurable DHF Structure
(Courtesy of Minerva)
Risk Management is a critical process: risks must be identified, evaluated, analyzed, and assessed to mitigate potential device issues. The Minerva solution captures risk assessments and mitigations as data elements that can be structured, searched, and reused. The data elements contain values, text, and links to other business objects in the solution such as Failure Mode and Effects Analysis (FMEA) capability shown in Figure 2, and change forms.
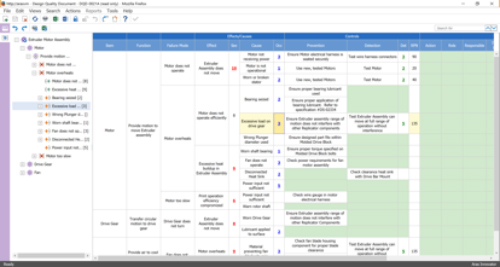
Figure 2—FMEA Accessed from Risk Assessment Form
(Courtesy of Minerva)
The Minerva Medical Device PLM solution also takes advantage of the commenting capability of the Aras’ Visual Collaboration solution shown in Figure 3. Ad hoc text, graphics, and threaded comments can be captured and associated to Aras business objects such as risk assessments. This capability takes discussions out of unmanaged or disconnected solutions like email and associates them with the product which they impact, helping capture the rationale behind decisions.
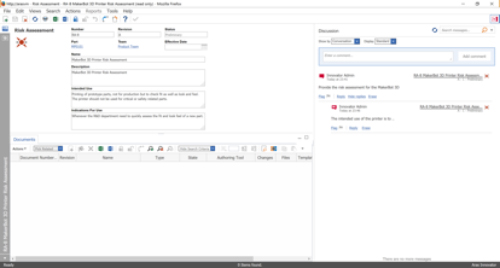
Figure 3—Risk Assessment Form Supports Collaboration and Commenting by Leveraging Aras’ Visual Collaboration Capabilities
(Courtesy of Minerva)
Companies interested in deploying the Minerva Medical Device PLM solution must also purchase a subscription from Aras Innovator. Subscribers get full access to Aras products including Requirements Management, Visual Collaboration, ECAD and MCAD integrations, and the IHS parts catalog integration, which is useful for products that contain electronic components. Minerva solutions are built using standard Aras configuration techniques so it is relatively easy to tailor the Minerva Medical Device PLM solution to leverage native Aras capabilities and support solution changes that add competitive advantages.
Minerva provides their own hotline for support and leverages Aras support when necessary. There is another significant benefit from being an Aras subscriber. The Minerva solutions are fully integrated into the Aras free database upgrade process, making it easy to evolve a deployment with new Aras Innovator releases. CIMdata has previously commented[1] on the value of this service, and sees it as a real differentiator.
Conclusion
Like all industries, medical device companies are facing increased pressure on time to market, cost, and quality. Legacy processes and systems based on file folders and even paper are common and negatively impact traceability, business efficiency, and of course risk. Accurate and complete DHF, DMR, and risk management records are critical to meeting regulatory requirements, but are difficult to support without a well-designed and implemented PLM solution.
The Minerva Medical Device PLM solution, built on the Aras Innovator PLM platform, configures Aras Innovator to support medical device specific data and processes based on industry best practices. This offers companies a faster way to get a PLM solution implemented. When compared to paper or file folder repositories, data linked together as a single source of truth is a huge value and competitive advantage. CIMdata finds this solution to be a great choice to get value quickly while still having the option to take advantage of the flexibility of Aras Innovator and the ecosystem surrounding it. Medical device companies looking to improve their processes and make better use of their product data should consider Minerva’s Medical Device PLM solution.




